There’s a lot riding on
your prostate health…
KNOW THE SCORE.
your prostate health…

ClarityDX Prostate uses clinical and blood-based biomarkers to determine a patient’s risk of having aggressive prostate cancer to help patients and their healthcare providers make more informed healthcare decisions.

Order ClarityDX Prostate Today
To order ClarityDX Prostate:
- Select the Province or Territory where you will get your blood sample collected from the options below.
- Download the Requisition Package and follow the instructions within.
- Pay for the test by selecting on-line payment (credit card) or offline payment (cheque or credit card).
Step 1
What Province Are You In?
*ClarityDX Prostate is currently available in Alberta, British Columbia, Ontario, Saskatchewan, Quebec and Yukon. If your province or territory is not listed, it is not available there yet. We are actively working with sample collection laboratories to expand availability across Canada.
PSA testing alone doesn’t add up
1 in 8 men will be diagnosed with prostate cancer in their lifetime. The good news is that when detected early, prostate cancer is one of the most successfully treated types of cancer (Sung et al., 2021).
The challenge is that the current method of screening – the prostate-specific antigen (PSA) test, looks at just one data point in a complex equation. The test measures levels of PSA, a protein made in the prostate, and flags elevated levels for risk of prostate cancer risk and further testing, including biopsy (Wilkinson et al., 2025).
Elevated PSA levels can indicate prostate cancer, but can also be caused by vigorous exercise, recent sexual activity, a prostate infection, etc. In fact, biopsies show most ‘positive’ PSA tests are not indicative of clinically significant prostate cancer on their own (McLaughlin et al., 2024; Van Poppel et al., 2022).
The result is that patients may opt for unnecessary, invasive prostate biopsies and treatments with potentially harmful side effects(Hyndman, Paproski et al., 2024; Hyndman, Kinnaird et al., 2024; Wallis et al., 2024).
Better outcomes start from clarity
When it comes to a cancer diagnosis, clarity means piece of mind. And when it comes to prostate cancer detection, it takes the right data to deliver that clarity.
ClarityDX Prostate is a clinically proven decision support tool that uses a machine learning platform to calculate an accurate prostate cancer risk score from clinical data and multiple blood-based PSA test results (Hyndman, Paproski et al., 2024; Hyndman, Kinnaird et al., 2024; Wallis et al., 2024).
If you have an elevated PSA, or a suspicion of prostate cancer, ClarityDX Prostate may be right for you. ClarityDX Prostate takes the fear and uncertainty out of prostate cancer screening.
Receiving your ClarityDX Prostate results
Your healthcare provider will receive the results and share them with you. You can also request access to your ClarityDX Prostate results. We will process your request and email your results to you via our secure email service.
Instructions:
- Click on the Access Record Form button and download the PDF document.
- Print and fill in page 1 of the form.
- Scan or take a photograph of the completed form.
- Scan or take a photograph of one (1) piece of photo ID (e.g. driver’s license, passport photo page, provincial ID card), or two (2) pieces of ID without a photo (e.g. provincial healthcare card, birth certificate, marriage certificate).
- Email a copy of the completed form and your ID photo (s) to Nanostics’ Privacy Officer (email address listed on the form).
The ClarityDX Prostate patient journey
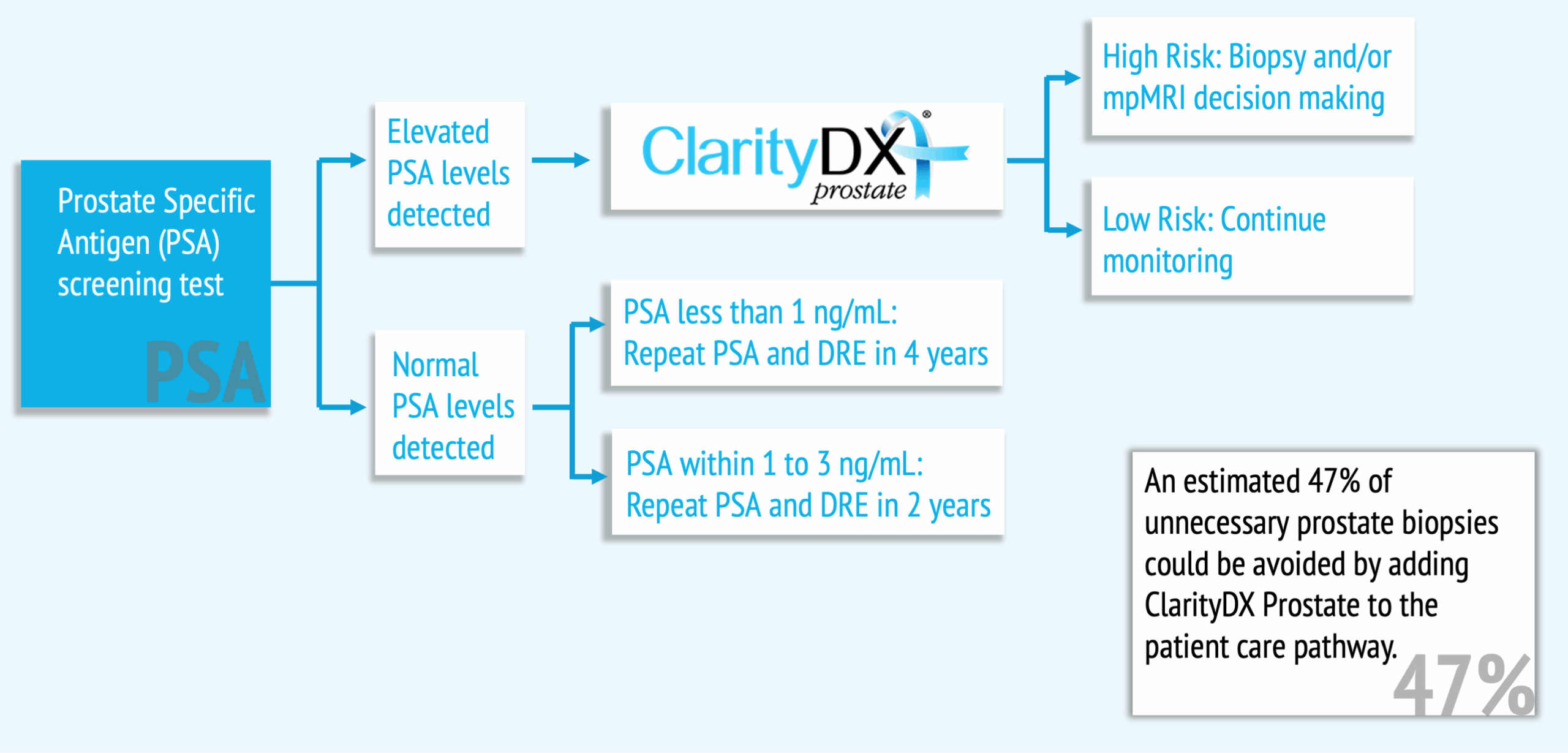
How does ClarityDX Prostate work?
ClarityDX Prostate uses a proprietary machine-learning algorithm and data from two blood-based biological markers with up to five clinical biomarkers to generate an accurate risk score for aggressive prostate cancer (Hyndman, Paproski et al., 2024; Hyndman, Kinnaird et al., 2024; Wallis et al., 2024).
Because ClarityDX Prostate uses lab data from a patient’s PSA blood test, it can be added to the healthcare pathway with minimal inconvenience to patients and low cost to the healthcare system.
ClarityDX Prostate can be utilized at multiple stages in the patient care path to inform the decision to biopsy.
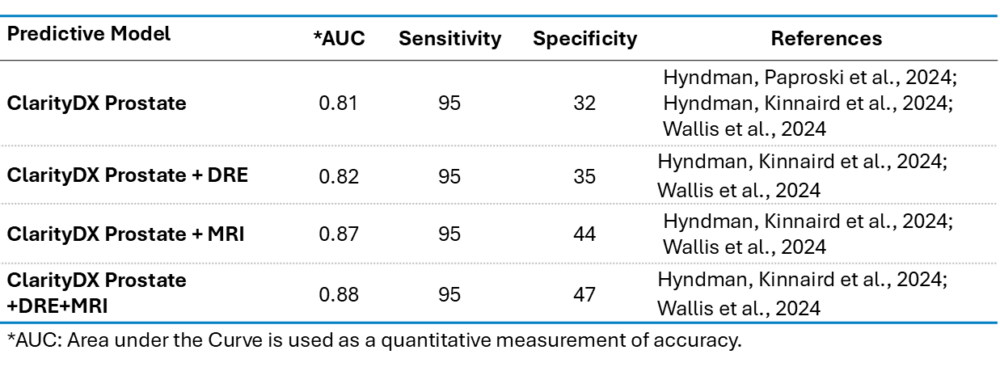
ClarityDX Prostate performance characteristics
Performance characteristics for ClarityDX Prostate models using up to five clinical features for predicting grade group 2 and above prostate cancer:
Frequently asked questions
Who is eligible for ClarityDX Prostate
Other FAQs:
References
1. Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA: A Cancer Journal for Clinicians 2021; 71(3): 209–49.
2. Wilkinson AN, Ellison LF, Zhang SX, et al. Canadian Prostate Cancer Trends in the Context of PSA Screening Guideline Changes. Current Oncology 2025; 32(12): 669.
3. McLaughlin PW, Cousins MM, Tsodikov A, Soni PD, Crook JM. Mortality reduction and cumulative excess incidence (CEI) in the prostate-specific antigen (PSA) screening era. Scientific Reports 2024; 14(1): 5810.
4. Van Poppel H, Albreht T, Basu P, Hogenhout R, Collen S, Roobol M. Serum PSA-based early detection of prostate cancer in Europe and globally: past, present and future. Nature Reviews Urology 2022; 19(9): 562–72.
5. Hyndman ME, Paproski RJ, Kinnaird A, et al. Development of an effective predictive screening tool for prostate cancer using the ClarityDX machine learning platform. npj Digital Medicine 2024; 7(1): 163.
6. Hyndman ME, Kinnaird A, Paproski RJ, et al. MP 1.13 Accurate risk models with and without magnetic resonance imaging features and digital rectal examination findings to predict clinically significant prostate cancer before prostate biopsy. Canadian Urological Association Journal 2024; 18(6S1): S21–9. Poster presented by Dr. CD Wallis, CUA 2024.
7. Wallis CJD, Paproski RJ, Hyndman ME, et al. Addition of MRI or DRE to clinical data and risk prediction for clinically significant prostate cancer. Journal of Clinical Oncology 2024; 42(16_suppl): e17105–e.
Disclaimer
The performance characteristics of ClarityDX Prostate were determined by Nanostics in a population primarily between 40 to 75 years of age with PSA ≥3 ng/mL. PSA and free PSA tests are indicated for men between ≥50 years of age; caution required when interpreting individual PSA and free PSA results in patients below 50 years of age. Accuracy may be influenced by PSA-altering drugs such as 5-alpha reductase inhibitors. Patient management should be based on holistic clinical judgment. ClarityDX Prostate has not been cleared or approved by the U.S. Food and Drug Administration (FDA) or Health Canada. ClarityDX Prostate is intended to aid physicians in the decision for subsequent diagnostic testing such as prostate biopsy or imaging.
CPSA accredited laboratory
Nanostics has an accredited laboratory at A108, 2011 94 Street, Edmonton, AB, Canada, T6N 1H1, under the College of Physicians and Surgeons of Alberta to perform total PSA, free PSA, and ClarityDX Prostate® measurements.



















