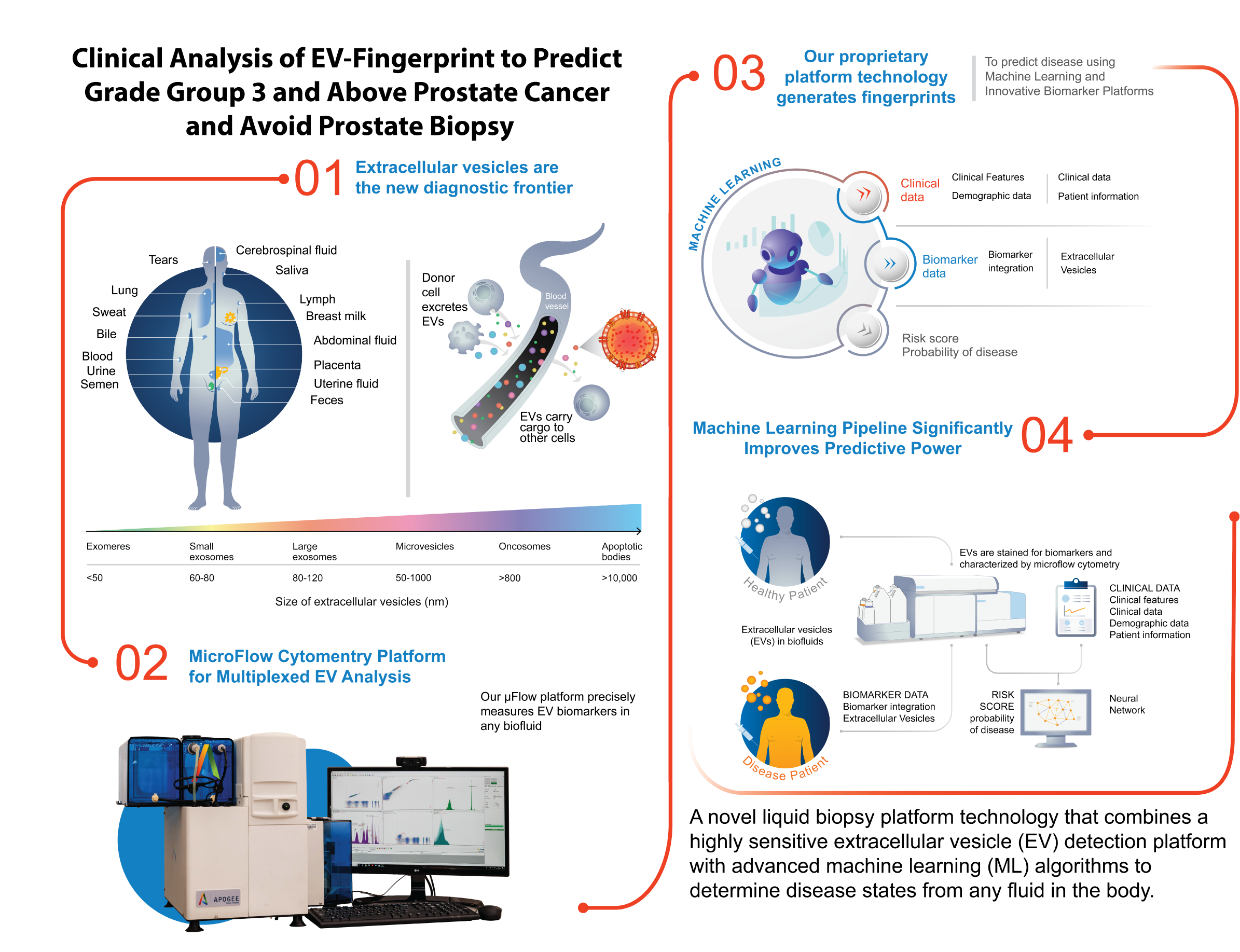
EV-Fingerprint test predicts aggressive prostate cancer
Clinical analysis of EV- Fingerprint to predict grade group 3 and above prostate cancer and avoid prostate biopsy
Adrian Fairey, Robert J. Paproski, Desmond Pink, Deborah L. Sosnowski, Catalina Vasquez, Bryan Donnelly, Eric Hyndman, Armen Aprikian, Adam Kinnaird, Perrin H. Beatty, John D. Lewis